The Adeno-Associated Viral vector is the safest and effective vehicle that is able to maintain long-term gene and protein expression. Adeno-associated viral vectors were developed back in the 1960s after some adenoviral vectors were found to be dangerous to patients.
 Gene Therapy Voyager Therapeutics
Gene Therapy Voyager Therapeutics
Key Highlights of AAV Vector-Based Gene Therapy Market.
Adeno associated virus gene therapy. The AAV genome is built of single-stranded deoxyribonucleic acid ssDNA either positive- or negative-sensed which is about 47 kilobase long. There has been a resurgence in gene therapy efforts that is partly fueled by the identification and understanding of new gene delivery vectors. It is a defective helper-dependent.
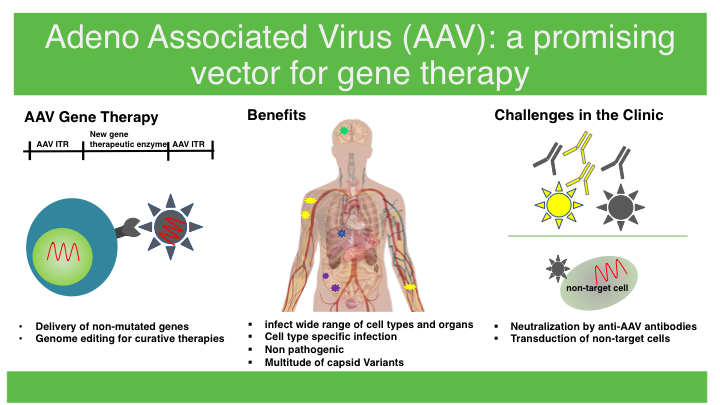
Adeno-associated virus AAV is a non-enveloped virus that can be engineered to deliver DNA to target cells and has attracted a significant amount of attention in the field especially in clinical-stage experimental therapeutic strategies. Adeno-associated virus AAV vector-mediated gene delivery was recently approved for the treatment of inherited blindness and spinal muscular atrophy and long-term therapeutic effects have been achieved for other rare diseases including haemophilia and Duchenne muscular dystrophy. Adeno-associated virus AAV based delivery techniques may hold the key.
Adeno-associated virus AAV vectors are the leading platform for gene delivery for the treatment of a variety of human diseases. They consist of single strands of DNA genomic material produced. Recent advances in developing clinically desirable AAV capsids.
Recent advances in developing clinically desirable AAV capsids optimizing genome designs and harnessing revolutionary biotechnologies have contributed substantially to the growth of the gene therapy field. This report delivers an in-depth understanding of the AAV Vector Based Gene Therapy historical and forecasted epidemiology as well as the AAV Vector Based Gene Therapy market trends in the United States EU5. Among these adeno-associated virus AAV has emerged as particularly promising because of its impressive safety profile and efficiency in transducing a wide range of cell types.
An international collaboration of leading groups in gene therapy and vision science have developed an adeno-associated virus AAV genome-coupled immunomodulation strategy that helps cloak the AAV virus from unwanted immune responses and offers important insights into ocular inflammation. Whereas AAV packages a single strand of DNA and must wait for its second strand to be synthesized scAAV packages two shorter strands that are complementary to each other. Genome organisation of Adeno-associated viruses.
Among the viral vectors that could be used for this purpose adeno-associated virus AAV has several features that make it particularly useful for gene therapy. The global trend for expansion continues with more than a thousand cell and gene therapy CGT clinical trials underway in 20191 Whether it is an introduction of genetic material into target cells in vivo or ex vivo cell therapy Adeno-Associated Viruses AAV have strengthened their position as a leading platform for gene delivery especially after recent landmark approvals. 2 The pathology manifests as mononuclear cell infiltrates and sensory neuron degeneration within the DRG in addition to secondary axonopathy which affects both the central axon of dorsal spinal cord SC tracts and the peripheral axon of peripheral nerves Fig.
Rep and cap see figure 1. Gene therapy using recombinant adeno-associated virus AAV has been linked with histopathological findings in dorsal root ganglion DRG sensory neurons in preclinical studies using nonhuman primates 1 and pigs. Adeno-associated virus AAV vectors are the leading platform for gene delivery for the treatment of a variety of human diseases.
Because of AAVs specialized gene therapy advantages researchers have created an altered version of AAV termed self-complementary adeno-associated virus scAAV. Adeno-Associated Virus AAV Vector Based Gene Therapy Understanding Gene therapy is a promising treatment option for a number of diseases including inherited disorders some types of cancer and. To date approximately two thirds of the 1800 gene therapy clinical trials completed worldwide have used viral vectors.
For adenoassociated virus AAVbased human gene therapy challenges for the translation of promising research results to successful clinical development include optimization of vector design and manufacturing processes to ensure that vectors prepared for administration to human subjects have attributes consistent with safe and durable expression. The genome comprises inverted terminal repeats ITRs at both ends of the DNA strand and two open reading frames ORFs. In the simplest form translating gene therapies from conceptual design to clinical trials involve identifying a therapeutic gene finding a means to deliver it and identifying a suitable route of administration.