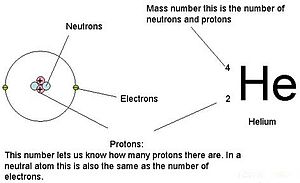
The atom has _____ electrons. This depiction shows the particles as separate whereas in an actual helium atom the protons are superimposed in space and most likely found at the very center of the nucleus and the same is true of the two neutrons.
The number of protons in the nucleus of an atom is its atomic number Z.
The number of protons in the nucleus of an atom. The atomic number is the number of protons in an atom of an element. An arrangement of the elements in order of their atomic numbers such that elements with similar properties fall in the same column. Try a smart search to find answers to similar questions.
Simplest type of a pure substance. The atomic number uniquely identifies a chemical element. It is identical to the charge number of the nucleus.
There are 19 protons and 21 neutrons in the nucleus. The mass of the atom is a unit called the atomic mass unit left textamu right. For example the atomic number of helium is two.
To counter this charge the atom will also contain 20 electrons. So in the event that you need to know the number of neutrons in uranium you should simply take away the atomic number 92 protons from the mass number 238 protons in addition to neutrons. In the nucleus the two protons and two neutrons are depicted in red and blue.
For example any atom that contains six protons is the element carbon and has the atomic number 6 regardless of how many neutrons or electrons it may have. Therefore there are 19 electrons in a potassium atom. Number of neutrons N 64 - 30 34.
C An atom has atomic mass number 23 and atomic number 11. B The total number of protons and neutrons in the nucleus of an atom is called _____. The interesting thing here is that every atom of krypton contains 36 protons.
Its value determines the identity of the atom. Number of protons Z 30. Uranium has an atomic number of 92 number of protons and a mass number of 238 protons in addition to neutrons.
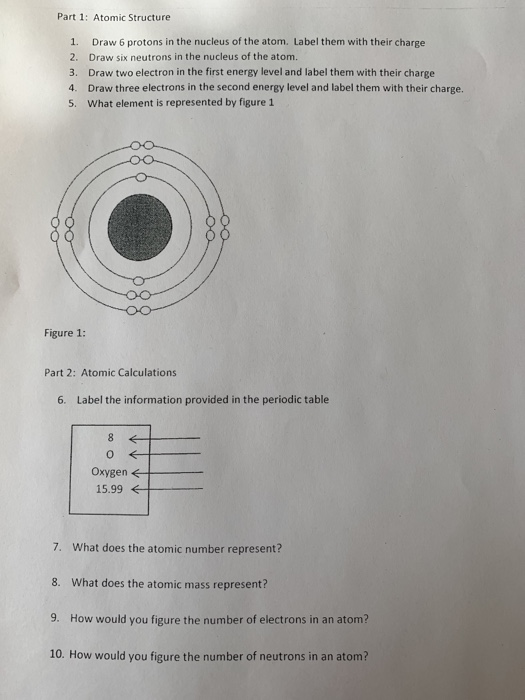
Number of protons in the nucleus of an atom page _ __f__ 7. Find an answer to your question In the nucleus of an atom there are 18 protons and 20 neutronsWhat are the atomic number and mass number of this isotope. A The number of protons in the nucleus of an atom is called its _____.
This is the defining trait of an element. In an uncharged atom the atomic number is also equal to the number of electrons. Protons are particles in the nucleus of an atom that have a positive charge equal to 1.
In Physics if the answers seem to be not correct or theres no answer. Step 1 - Gather Information. The nucleus of isotope A of an element has a larger mass than isotope B of the same element.
For example boron B has an atomic number of 5 therefore it has 5 protons and 5 electrons. For hydrogen the number of protons is 1. Lithium is one example where the number of neutrons is not equal to the number of protons.
Protons are important because they define what element an atom is from. One atomic mass unit is the mass of a proton or about 167 times 10-27 kilograms which is an extremely small mass. The atomic number determines the chemical properties of the atom.
The number of PROTONS in the nucleus of isotope A is _____ the number of PROTONS in the nucleus of isotope B. The element of an atom with 2 protons is always helium. There are 30 protons and 34 neutrons.
For this reason chemical elements are represented by the number of protons in a nucleus Z that is the atomic number. The mass number A of an atom is the total number of protons and neutrons in its nucleus. The number of protons within the nucleus of a given atom is equal to the atomic number of the corresponding element which can be found on the periodic table.
Since protons and neutrons have approximately the same mass and the mass defe. Page _ o 9an abbreviation for the elements name page _ l 10. Atom of an element that has the same number of protons as another atom of the same element but a different number of neutrons.
D An atom of an element has 11 protons 11 electrons and 12 neutrons. The weighted average of the masses of all the naturally. This tells us that an atom of krypton has 36 protons in its nucleus.
Hence the atom will contain a total of 20 protons in its nucleus. The number of electrons is equal to the number of protons. For lighter elements such as helium carbon and oxygen it is common for the number of protons to be equal to the number of neutrons.
Total number of protons and neutrons in an atoms nucleus page _ __h__ 8. For zinc the number of protons is 30. If you are given the atomic weight of an atom you need to subtract the number of neutrons to get the number of protons.
The sum of the atomic number Z and the number of neutrons N gives the mass number A of an atom. The atomic number or proton number of a chemical element is the number of protons found in the nucleus of every atom of that element. Charged particle formed when an atom gains or loses electrons.
Note that there are no electrons inside the nucleus electrons. Since this is a nucleus there are no electrons. In our example kryptons atomic number is 36.
Table gives the number of protons number of neutrons and the nuclide notations of some common elements. The atomic number of an atom is the number of protons in its nucleus. Therefore an element in a neutral state will have the same number of protons and electrons.
Number of protons in the nucleus of an atom. Electrons are particles that have a negative charge equal to -1. Number of protons neutrons A 64.
Therefore the number of protons is also two Explanation.